Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
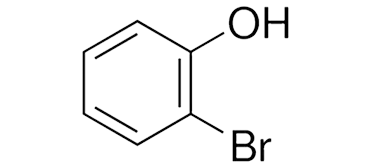

2-BromoPhenol is a halogenated aromatic compound widely recognized for its role in organic synthesis and pharmaceutical intermediate production. Known for its distinct structural features and reactivity, 2-BromoPhenol serves as an essential building block in a variety of chemical applications. In this article, we explore the properties, preparation methods, and industrial uses of 2-BromoPhenol, as well as safety measures necessary for its handling and storage.
2-BromoPhenol is an organic compound that belongs to the phenol family, featuring a hydroxyl group (-OH) attached to a benzene ring substituted with a bromine atom in the ortho position. Its molecular formula is C6H5BrO, and it appears as a pale to light yellow solid with a sharp, phenolic odor.
Molecular Formula: C₆H₅BrO
Molecular Weight: 173.01 g/mol
IUPAC Name: 2-Bromophenol
Melting Point: 34–36 °C
Boiling Point: Approximately 205–210 °C
Solubility: Slightly soluble in water; soluble in organic solvents like ethanol and ether
This compound is particularly valued for its reactivity, making it a useful intermediate in the synthesis of more complex molecules.
2-BromoPhenol is typically synthesized through electrophilic aromatic substitution. A common route involves the bromination of phenol under controlled conditions to selectively introduce the bromine atom at the ortho position. The process can be fine-tuned to achieve high yields and minimize side products.
In laboratory and industrial settings, bromination is carried out using bromine (Br₂) or N-bromosuccinimide (NBS) in the presence of a suitable solvent such as acetic acid or carbon tetrachloride.
One of the primary uses of 2-BromoPhenol is as an intermediate in the synthesis of pharmaceuticals. It serves as a key precursor in the manufacture of bioactive molecules, particularly in the development of anti-inflammatory and antimicrobial drugs. Its bromine group allows for further functionalization through cross-coupling and substitution reactions.
In the agricultural sector, 2-BromoPhenol is employed in the production of fungicides and herbicides. Its ability to be modified into more complex compounds makes it a versatile starting material for agrochemical formulations.
2-BromoPhenol is frequently used in organic synthesis due to its functional groups that can undergo various transformations, including Suzuki, Heck, and Sonogashira coupling reactions. These reactions enable chemists to create elaborate molecular frameworks required in material science and medicinal chemistry.
Some industrial dyes and pigments are derived from halogenated phenols such as 2-BromoPhenol. Its chemical properties allow it to react with aromatic and heterocyclic systems, forming the basis for colorants used in textiles, printing inks, and plastics.
Specialty polymers and resins sometimes incorporate 2-BromoPhenol as a reactive monomer. Its phenolic group can participate in polymerization reactions, while the bromine atom offers points for additional chemical modification.
Like many halogenated organic compounds, 2-BromoPhenol requires careful handling due to its potential health hazards.
May cause skin and eye irritation upon contact
Can be harmful if inhaled or ingested
Prolonged exposure may result in respiratory or dermal sensitization
Always use personal protective equipment (PPE) including gloves, goggles, and lab coats
Operate in a well-ventilated area or fume hood to avoid inhalation of vapors
Store in tightly sealed containers, away from moisture, heat, and light
Disposal of 2-BromoPhenol and its waste materials should be carried out in accordance with local environmental regulations. It is recommended to consult material safety data sheets (MSDS) and involve hazardous waste disposal services for proper management.
Store in a cool, dry place, away from incompatible substances such as strong oxidizing agents
Use non-reactive containers and ensure labels are clear and up to date
Keep away from direct sunlight and moisture to preserve stability
2-BromoPhenol plays a significant role in both industrial and research-oriented chemical processes. Its unique combination of a hydroxyl group and a bromine substituent allows for wide-ranging applications, from pharmaceutical synthesis to polymer production. While this compound offers valuable functionality, appropriate safety and handling measures must be observed to ensure secure usage. Chloromethyl methyl ether is a versatile reagent widely used in the protection of hydroxyl groups during organic synthesis.
By understanding the structure, behavior, and reactivity of 2-BromoPhenol, chemists and manufacturers can leverage its full potential while maintaining safety and regulatory compliance.